Tesamorelin
Meet your supplier

Distributor / Wholesaler • 3 yr •  UNITED KINGDOM
UNITED KINGDOM
56%
On-time delivery rate
≤24h
Response Time
75
Employee Size
$500K
Annual Revenue
588
Products Sold
Location
 Belfast, United Kingdom
Belfast, United Kingdom
KEY FEATURES
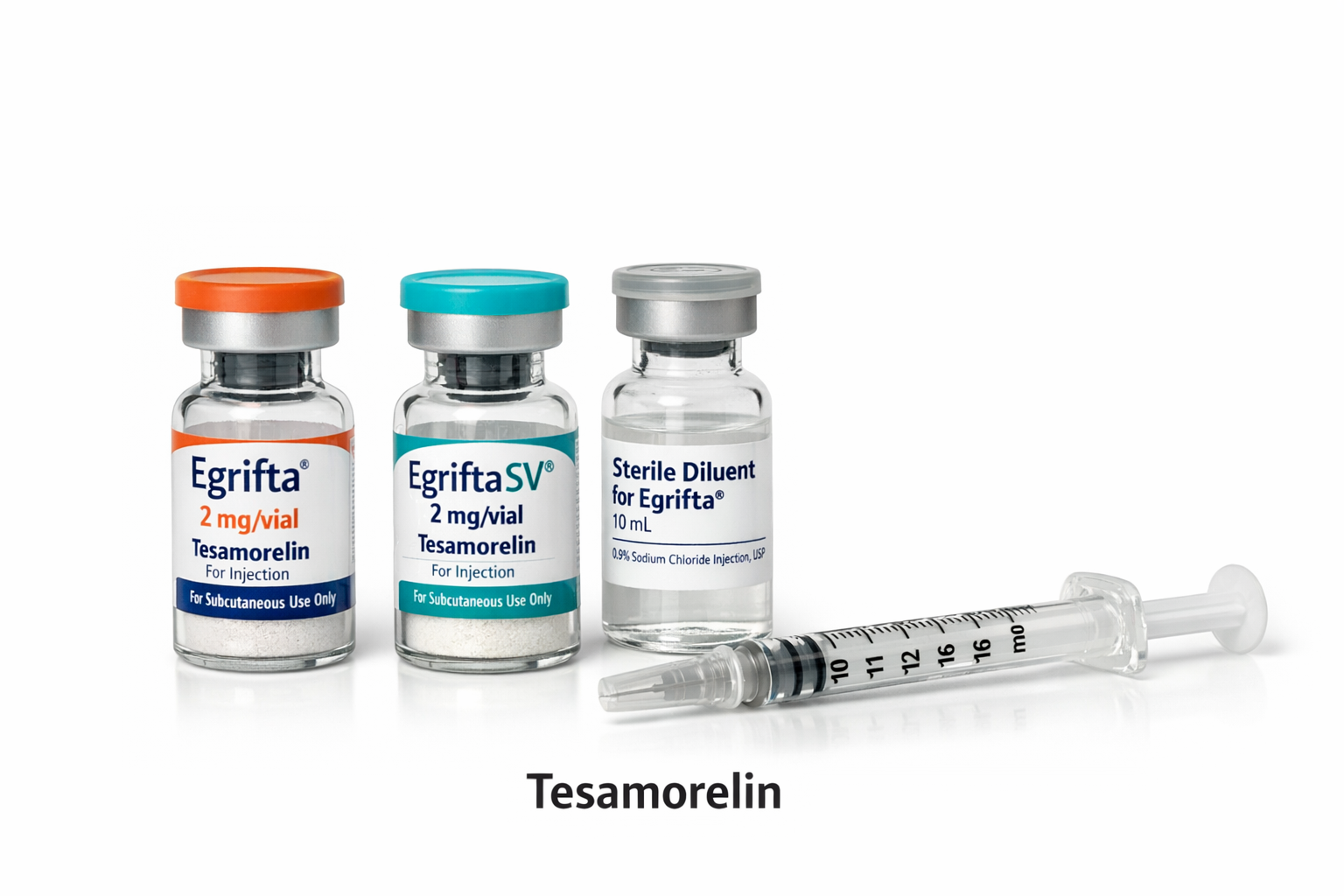
Therapeutic Class: Synthetic growth hormone–releasing factor (GRF) analog
Primary Indication: Reduction of excess abdominal fat in adults with HIV‑associated lipodystrophy
Mechanism of Action: Stimulates pituitary growth hormone release → increases IGF‑1 → reduces visceral adipose tissue
Clinical Benefit: Proven reduction in abdominal fat without affecting limb or subcutaneous fat distribution
Brand Names: Egrifta®, Egrifta SV®
TECHNICAL SPECIFICATIONS
Molecular Type: Synthetic peptide (44‑amino acid analog of human GRF)
Formulation: Sterile lyophilized powder for reconstitution
Dosage strength
Egrifta®: 1 mg and 2 mg vials
Egrifta SV®: 2 mg vials (small‑volume, more concentrated)
Administration: Daily subcutaneous injection into abdominal area
Preparation: Reconstituted with supplied sterile diluent before injection
Storage: Refrigerated at 2–8 °C; do not freeze; use reconstituted solution immediately
Regulatory Identifiers: Each vial carries an NDC code (U.S.) or equivalent international code for compliance
AVAILABLE CONFIGURATIONS
Format and Delivery:
Lyophilized powder vials (single‑use)
Supplied with diluent for reconstitution
Concerntration and Dosage:
Standard Egrifta® vials (1 mg, 2 mg)
Egrifta SV® vials (2 mg, reduced diluent volume)
Packaging and Labeling:
Cartons containing vials + diluent
Professional labeling with regulatory compliance identifiers