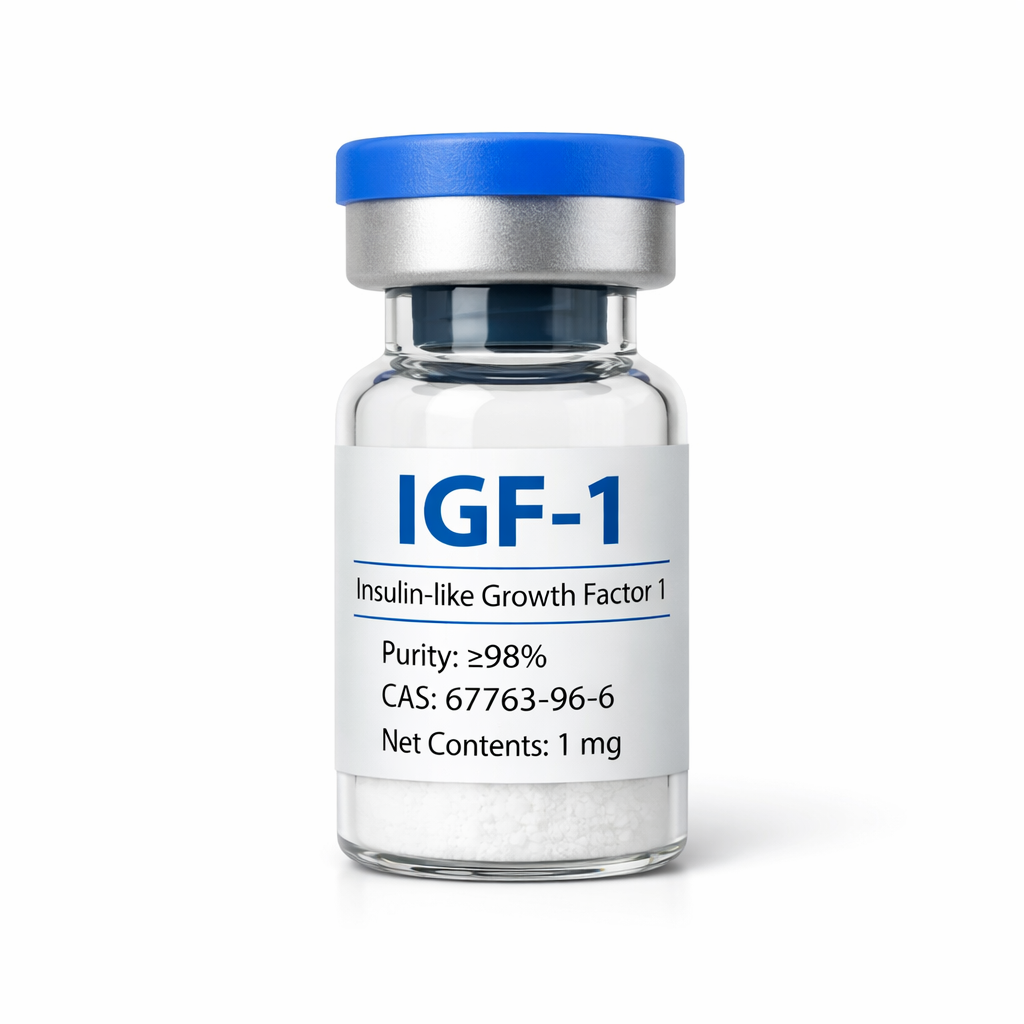
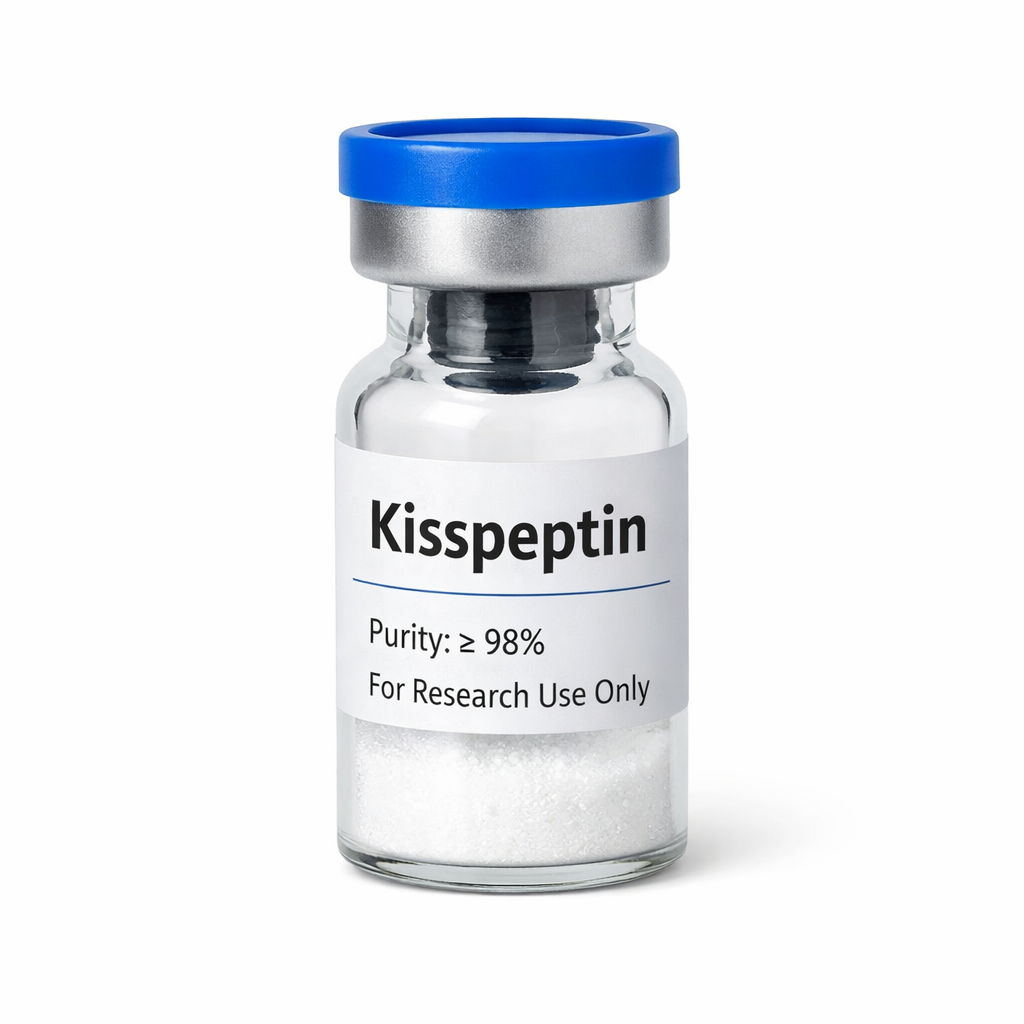
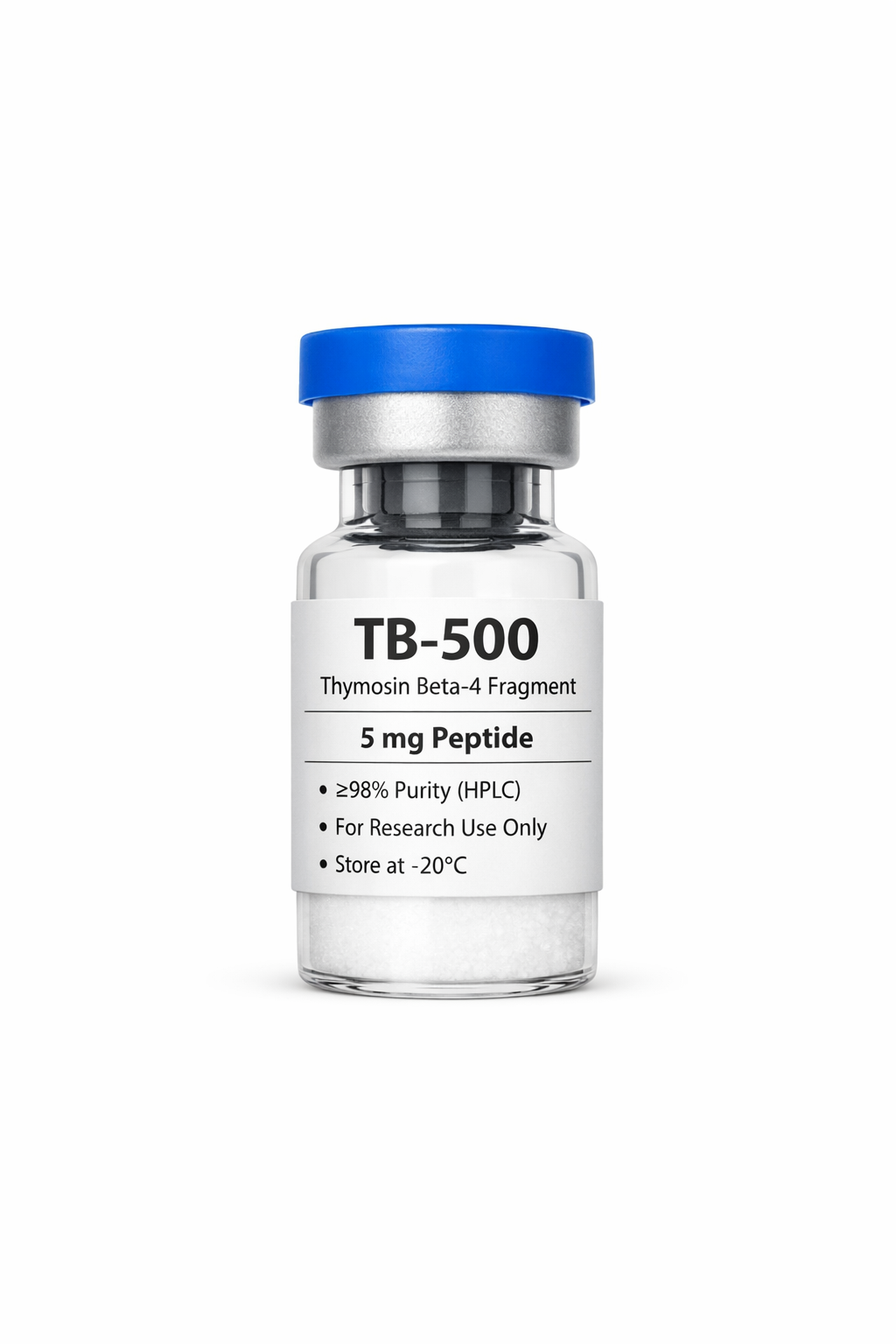
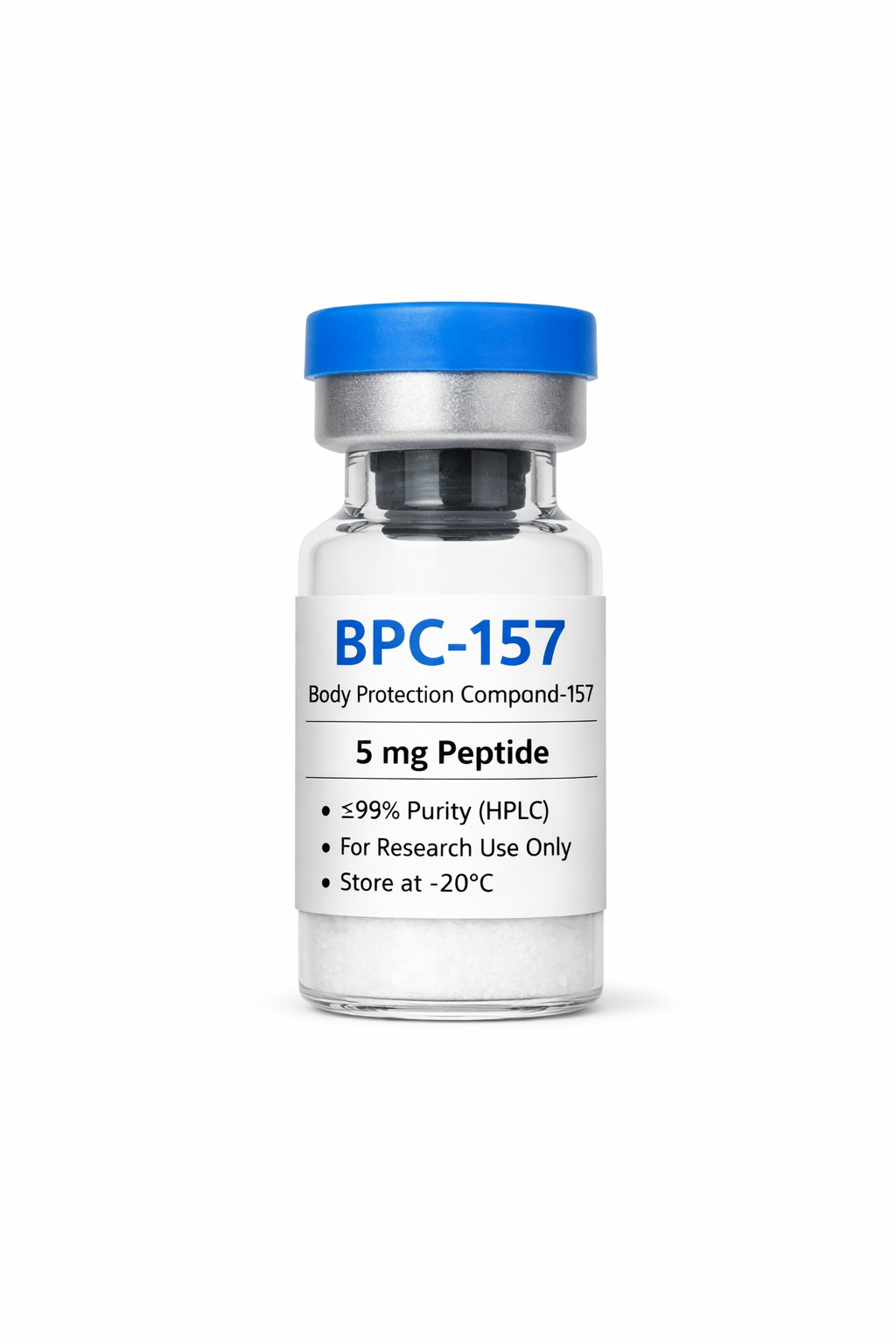
Sema~Glutide
Meet your supplier

Distributor / Wholesaler • 3 yr •  UNITED KINGDOM
UNITED KINGDOM
56%
On-time delivery rate
≤24h
Response Time
75
Employee Size
$500K
Annual Revenue
588
Products Sold
Location
 Belfast, United Kingdom
Belfast, United Kingdom
KEY FEATURES
Therapeutic Class: GLP‑1 receptor agonist (peptide therapy)
Primary Uses: Type 2 diabetes management, chronic weight management
Mechanism of Action :
Stimulates insulin secretion when glucose is elevated
Reduces glucagon release
Slows gastric emptying → appetite suppression
Clinical Benefits :
Proven HbA1c reduction
Average 10-15% weight loss during trial
Cardiovascular protection (reduced risk of heart attack/stroke in diabetic patients)
TECHNICAL SPECIFICATIONS
Molecular Type: Synthetic peptide (semaglutide)
Dosage forms :
Injection pens: 0.25 mg, 0.5 mg, 1 mg, 2 mg (weekly dosing)
Oral tablets: 3 mg, 7 mg, 14 mg (daily dosing)
Brands/Models:
Ozempic® – Weekly injection pen for type 2 diabetes
Wegovy® – Weekly injection pen for weight management
Rybelsus® – Daily oral tablet for type 2 diabetes
Storage & Handling :
Injection pens: Refrigerate until first use; room temperature up to 56 days after opening
Tablets: Store at room temperature, away from moisture
Safety profile :
Common side effects: Nausea, diarrhea, constipation
Serious risks: Pancreatitis, gallbladder issues, kidney problems, rare thyroid tumors
Contraindications: Type 1 diabetes, pregnancy, breastfeeding, history of medullary thyroid carcinoma or MEN2
AVAILABLE CONFIGURATIONS
Format and Delivery :
Prefilled injection pens (fixed strengths)
Oral tablets (daily dosing)
Compounded vials (for specialized clinical supply)
Concerntration & Dosing:
Branded pens: fixed strengths (0.25–2 mg)
Oral tablets: fixed strengths (3–14 mg)
Custom compounding: adjustable concentrations for research or clinical customization
Packaging & Lebeling:
Bulk API shipments
Finished product packaging (retail-ready)
Private labeling / white‑label options for distributors
Shipping and Compliance :
Cold chain logistics available
Flexible Incoterms (FOB, CIF, DAP, DDP)
Documentation tailored to HS code classification and regulatory requirements