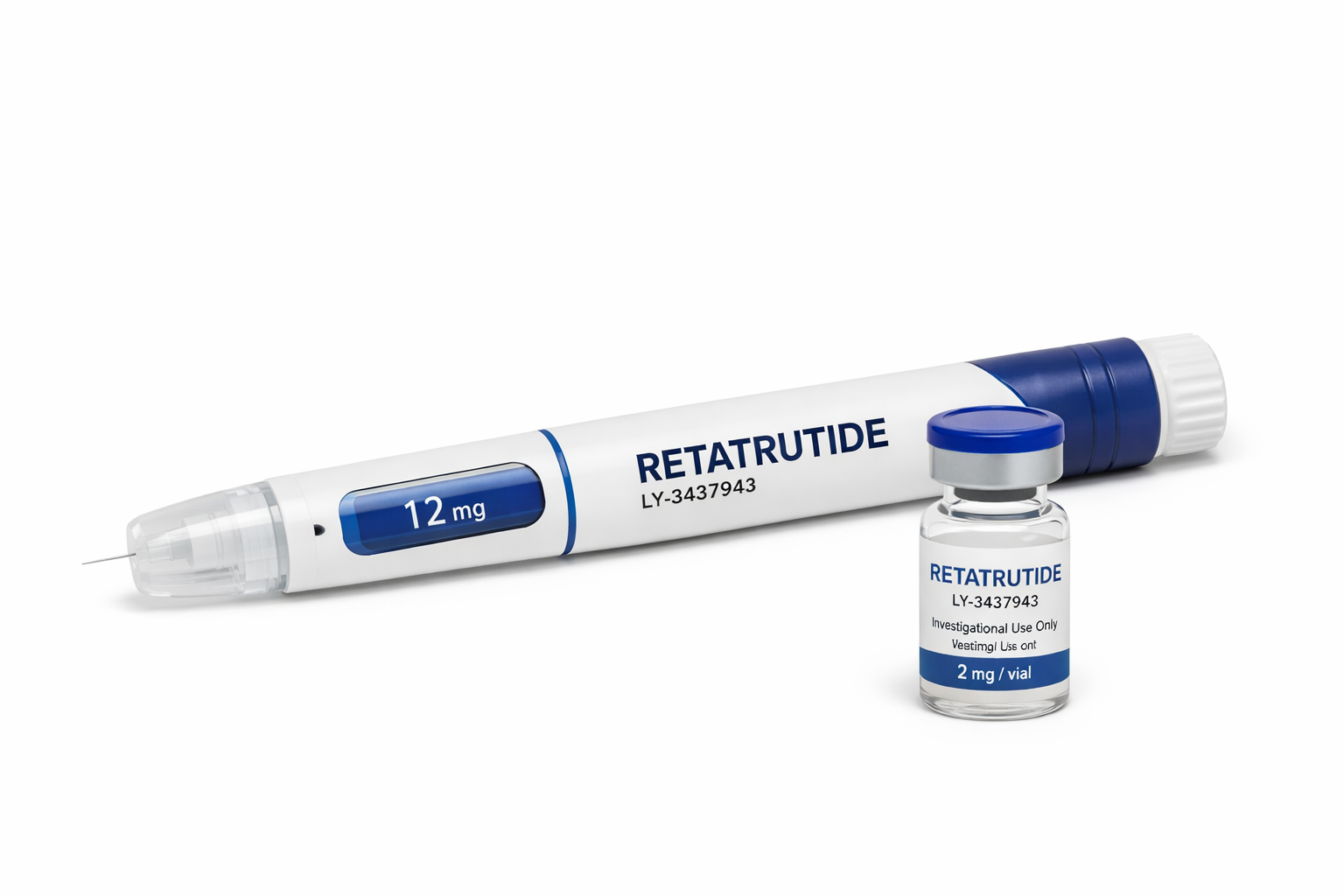
Reta~trutide
Meet your supplier

Distributor / Wholesaler • 3 yr •  UNITED KINGDOM
UNITED KINGDOM
56%
On-time delivery rate
≤24h
Response Time
75
Employee Size
$500K
Annual Revenue
588
Products Sold
Location
 Belfast, United Kingdom
Belfast, United Kingdom
KEY FEATURES
Therapeutic Class: Investigational peptide therapy
Mechanism of Action: Triple receptor agonist (GIP, GLP‑1, glucagon)
Primary Uses (Investigational): Obesity management, metabolic regulation, type 2 diabetes (under study)
Clinical Benefits (Trial Data):
Up to 24–25% body weight reduction in <1 year
Improved glucose metabolism and insulin sensitivity
Appetite suppression and increased energy expenditure
Status: Not FDA‑approved; currently in Phase II/III clinical trials
TECHNICAL SPECIFICATIONS
Molecular Type: Synthetic peptide (LY‑3437943)
Administration Route: Subcutaneous injection (once weekly)
Formulation: Prefilled injection pens (trial use)
Stability & Storage: Refrigerated until use; stability data under investigation
Safety Profile (Investigational):
GI side effects (nausea, diarrhea)
Risks under study: bone health, kidney complications, rapid weight loss
Regulatory Identifiers: Not yet assigned ATC code or HS classification (pending approval)
AVAILABLE CONFIGURATIONS
Format & Delivery:
Prefilled injection pens (trial formulations)
Bulk API (research supply only)
Concentration & Dosing:
Investigational fixed strengths (dose escalation in trials)
No commercial dosing yet established
Packaging & Labeling:
Clinical trial packaging only
Future potential: finished product packaging, private labeling (post‑approval)